Red and near-infrared light therapy shows remarkable therapeutic potential, but emerging evidence suggests that melanin content fundamentally alters how tissues respond to photobiomodulation. Understanding these interactions could revolutionize personalized light-based medicine and reveal new dimensions of melanin's biological significance.
The wavelengths that heal may not be universal. When dermatologist Endre Mester first discovered that low-level laser light could accelerate wound healing in mice back in 1967, he unknowingly opened a door to one of medicine's most promising yet puzzling therapeutic modalities. Today, photobiomodulation therapy (PBMT) using red (660-670 nm) and near-infrared (810-850 nm) light has demonstrated efficacy across dozens of conditions, from chronic pain to neurodegeneration. Yet a critical variable has been largely overlooked: the profound influence of melanin on how these therapeutic wavelengths interact with living tissue.
Recent investigations into melanin's optical properties reveal that this ancient pigment doesn't simply absorb light uniformly across the spectrum. Instead, melanin exhibits complex wavelength-dependent behaviors that could explain why photobiomodulation outcomes vary so dramatically between individuals—and why the field has struggled to establish consistent dosing protocols.
The Cytochrome Connection: Primary Targets of Therapeutic Light
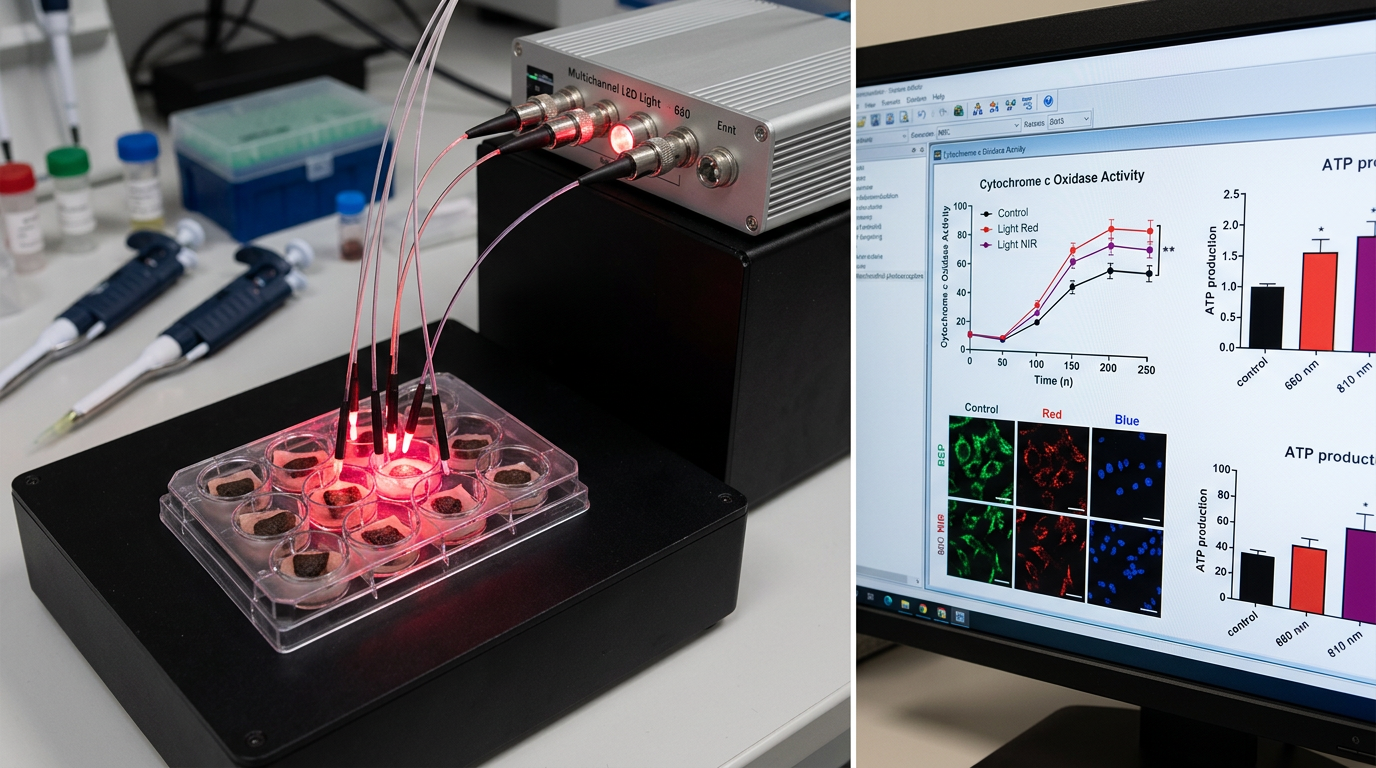
The established mechanism of photobiomodulation centers on cytochrome c oxidase (CCO), the terminal enzyme in the mitochondrial electron transport chain. This copper-containing protein absorbs specific wavelengths of red and near-infrared light, leading to increased ATP production, enhanced cellular respiration, and cascading effects on cellular signaling pathways.
Tiina Karu's pioneering work at Moscow State University demonstrated that CCO's absorption spectrum closely matches the therapeutic windows used in PBMT, with peaks around 665 nm, 810 nm, and 880 nm. When photons of these energies strike CCO, they can dissociate inhibitory nitric oxide from the enzyme's active site, effectively removing the molecular "brake" on cellular energy production.
However, this elegant mechanism assumes that therapeutic light actually reaches mitochondria-rich tissues. In melanin-rich skin, this assumption requires careful examination. Eumelanin, the brown-black pigment predominant in darker skin types, exhibits broadband absorption across the visible and near-infrared spectrum, with absorption coefficients that can exceed 1000 cm⁻¹ at therapeutic wavelengths.
The implications are profound: while a 810 nm photon might penetrate several centimeters into lightly pigmented tissue to reach muscle or neural targets, the same photon may be absorbed within millimeters in highly melanized skin. This isn't simply a matter of "turning up the power"—melanin's interaction with light involves complex photochemical processes that may generate their own biological effects.
Melanin as Photoacceptor: Beyond Simple Absorption
Traditional models treat melanin as an optical filter—a biological sunscreen that uniformly attenuates incoming light. Recent biophysical research reveals a far more sophisticated picture. Melanin granules, particularly in their hydrated state within living cells, demonstrate semiconductor-like properties with a bandgap of approximately 1.85 eV, corresponding to wavelengths around 670 nm.
This semiconductor behavior suggests that melanin doesn't merely absorb photons but can undergo photoexcitation, generating mobile charge carriers and potentially serving as a biological photovoltaic system. Work by Arturo Solís Herrera has proposed that melanin can split water molecules when exposed to light, generating hydrogen and oxygen in a process analogous to photosynthesis.
While these mechanisms remain under investigation, they point to a crucial insight: melanin-rich tissues may respond to photobiomodulation through entirely different pathways than those mediated by cytochrome c oxidase. Rather than competing with CCO for photons, melanin may serve as an additional—or alternative—photoacceptor system.
Consider the implications for therapeutic dosing. Current PBMT protocols typically recommend fluences (energy densities) of 1-10 J/cm² based on studies conducted primarily on cell cultures or animal models with minimal melanin content. But if melanin generates its own photobiological responses, optimal dosing for highly pigmented tissues might follow completely different dose-response curves.
Spectral Selectivity and Tissue Penetration Dynamics
The interaction between therapeutic light and melanin varies dramatically across the photobiomodulation spectrum. At 660 nm, near melanin's semiconductor bandgap, absorption is maximal—potentially triggering melanin-specific photochemical processes while limiting penetration depth. At 810 nm, melanin absorption decreases somewhat, allowing greater tissue penetration but still representing a significant optical barrier compared to unpigmented tissue.
Recent Monte Carlo modeling studies have quantified these differences. In skin with high melanin content (Fitzpatrick types V-VI), 660 nm light shows 90% attenuation within the first 0.5 mm of tissue. In contrast, the same wavelength penetrates 2-3 mm in lightly pigmented skin before reaching equivalent attenuation levels.
This spectral selectivity creates a therapeutic paradox. The wavelengths that most effectively activate cytochrome c oxidase in deeper tissues may be precisely those most strongly absorbed by superficial melanin. Conversely, wavelengths that penetrate melanin-rich skin more effectively may be suboptimal for mitochondrial photoacceptors.
The solution may lie in understanding melanin's own therapeutic potential. If melanin-mediated photobiomodulation operates through distinct mechanisms—perhaps involving proton conductivity, free radical modulation, or bioelectric signaling—then superficial melanin activation might generate therapeutic effects that complement or even surpass those achieved through deep tissue CCO stimulation.
Implications for Personalized Photomedicine
These findings demand a fundamental reconsideration of photobiomodulation protocols. Rather than applying universal dosing parameters, effective PBMT may require melanin-adjusted approaches that account for individual pigmentation levels, anatomical location, and therapeutic targets.
For highly melanized tissues, this might mean employing longer wavelengths (900-1000 nm) that penetrate more effectively, accepting lower CCO activation in favor of greater tissue penetration. Alternatively, it might involve embracing melanin as a therapeutic target itself, using wavelengths optimized for melanin photoexcitation rather than mitochondrial activation.
The clinical implications extend beyond dermatology. Neurological applications of photobiomodulation—from traumatic brain injury treatment to Parkinson's disease therapy—must account for the substantial melanin content in brain regions like the substantia nigra. Here, neuromelanin may serve as both a therapeutic target and a barrier to deeper tissue penetration.
Key Takeaways
• Melanin's semiconductor properties and broadband absorption fundamentally alter how red and near-infrared light penetrates and affects pigmented tissues, challenging universal photobiomodulation protocols.
• The established cytochrome c oxidase mechanism of photobiomodulation may be supplemented or replaced by melanin-specific photochemical pathways in highly pigmented tissues.
• Wavelength selection for photobiomodulation therapy should consider melanin content, with 660 nm light maximally absorbed by melanin's semiconductor bandgap and longer wavelengths offering better penetration.
• Current therapeutic dosing recommendations, derived primarily from studies on lightly pigmented subjects, may be inadequate or inappropriate for individuals with high melanin content.
• Neurological applications of photobiomodulation must account for neuromelanin in brain regions like the substantia nigra, where pigmentation may influence therapeutic outcomes.
• Personalized photomedicine approaches that adjust wavelength, fluence, and treatment protocols based on individual melanin content may optimize therapeutic efficacy across diverse populations.
References
Karu, T. "Primary and secondary mechanisms of action of visible to near-IR radiation on cells." Journal of Photochemistry and Photobiology B 49(1), 1-17 (1999).
Hamblin, M.R. "Mechanisms and applications of the anti-inflammatory effects of photobiomodulation." AIMS Biophysics 4(3), 337-361 (2017).
McGinness, J., Corry, P., & Proctor, P. "Amorphous semiconductor switching in melanins." Science 183(4127), 853-855 (1974).
Meredith, P. & Sarna, T. "The physical and chemical properties of eumelanin." Pigment Cell Research 19(6), 572-594 (2006).
Solís-Herrera, A., Arias-Esparza, M.C., & Solís-Arias, R.I. "The unexpected capacity of melanin to dissociate the water molecule fills the gap between the life before and after ATP." Biomedical Research 21(2), 224-226 (2010).
Zecca, L., Youdim, M.B., Riederer, P., Connor, J.R., & Crichton, R.R. "Iron, brain ageing and neurodegenerative disorders." Nature Reviews Neuroscience 5(11), 863-873 (2004).
Tedford, C.E., DeLapp, S., Jacques, S., & Anders, J. "Quantitative analysis of transcranial and intraparenchymal light penetration in human cadaver brain tissue." Lasers in Surgery and Medicine 47(4), 312-322 (2015).